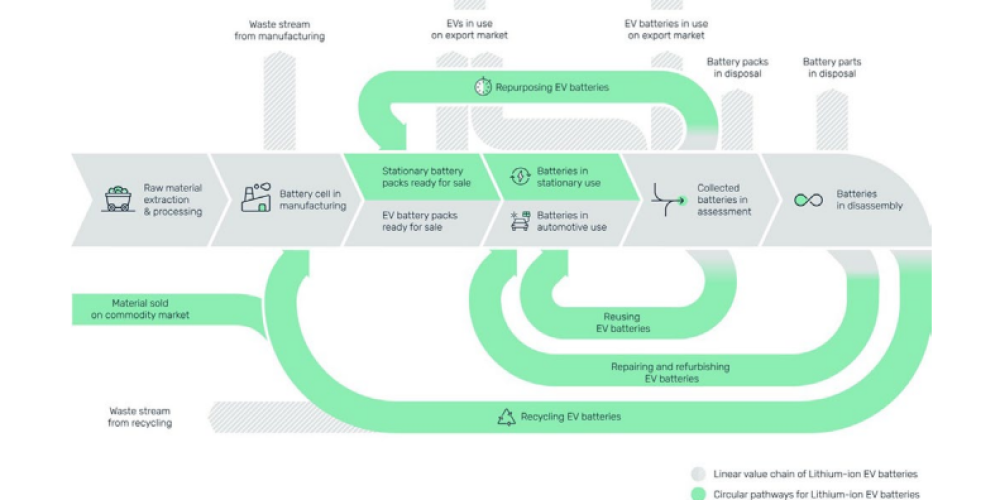
A New Approach to Battery Raw Materials in Switzerland. The energy transition and the shift to electromobility are bringing energy storage into focus globally for industry, politics, and society. In the coming years, the demand for decentralized energy storage will skyrocket worldwide. Consequently, the demand for the required raw materials will also grow. FESS_Compendium_2_2023
On the afternoon of May 4, 2023, the ESM Foundation hosted a momentous symposium to honor and celebrate the retirement of their esteemed colleague on the Board of Trustees, Prof. Dr. Armin Reller. This symposium served as a platform for reflecting upon Armin Reller’s illustrious professional journey and paying tribute to his exceptional contributions as a visionary scientist in the realm of forward-thinking resource research. Building upon the groundbreaking concept of the “stories of stuff”, a concept pioneered by Armin Reller himself, which delves into the origins and destinies of materials and resources, the occasion provided an opportunity to contemplate the past, present, and future. Through an engaging symposium, we […]
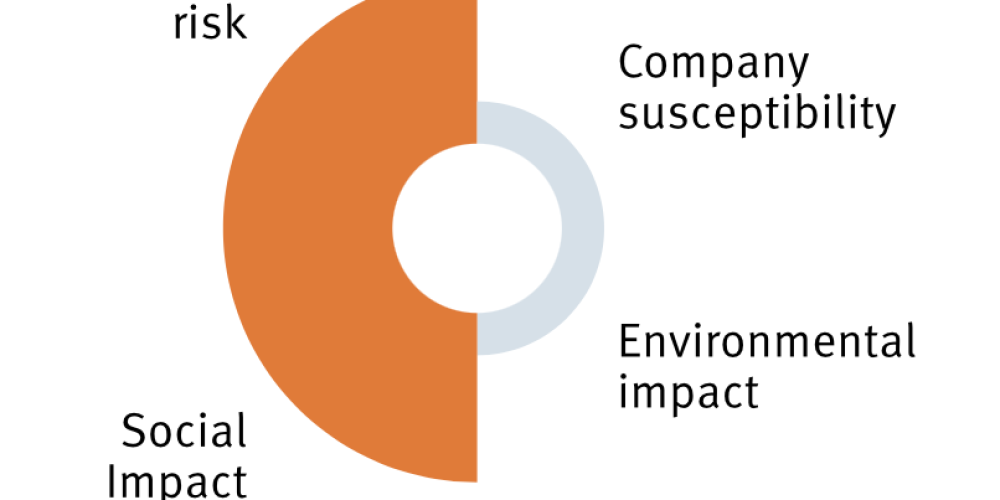
The European Commission has just presented the «Critical Raw Materials Act» to ensure the supply of critical raw materials in Europe and to reduce dependencies (LINK). Do you know which raw materials and semi-finished products are critical for your company’s operation and could lead to supply bottlenecks? The «Metal Risk Check» online tool developed by Swissmem, Empa and EBP enables an initial assessment of whether a company is exposed to supply risks from metals, the environmental pollution and social impacts associated with them and how to deal with them proactively. Thanks to the support of the ESM Foundation and the Federal Office for the Environment (FOEN), a new version of […]
The struggle to access copper, lithium, nickel and others, the new oil, is intensifying. Between lack of know-how, local opposition and logistical challenges, Europe is lagging behind. There were reactions in 2022, but not in Switzerland https://www.letemps.ch/economie/tres-gros-defis-minerais-transition
For an issue on raw materials of the Swiss magazine “Swissfuture – Magazin für Zukünfte” in December 2022, the ESM wrote an article on “Critical raw materials: a topic for the Switzerland of the future”. You can download the magazine here (in German): https://www.swissfuture.ch/wp-content/uploads/2022/12/sf_rohstoffe_02-22.pdf